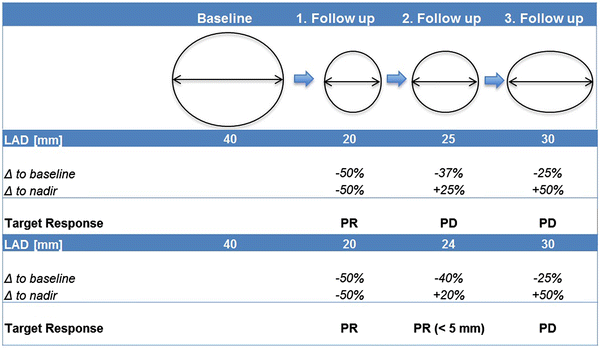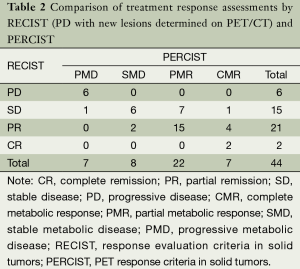
PET/CT evaluation of response to chemotherapy in non-small cell lung cancer: PET response criteria in solid tumors (PERCIST) versus response evaluation criteria in solid tumors (RECIST) - Ding - Journal of Thoracic

Validity of RECIST Version 1.1 for Response Assessment in Metastatic Cancer: A Prospective, Multireader Study | Radiology
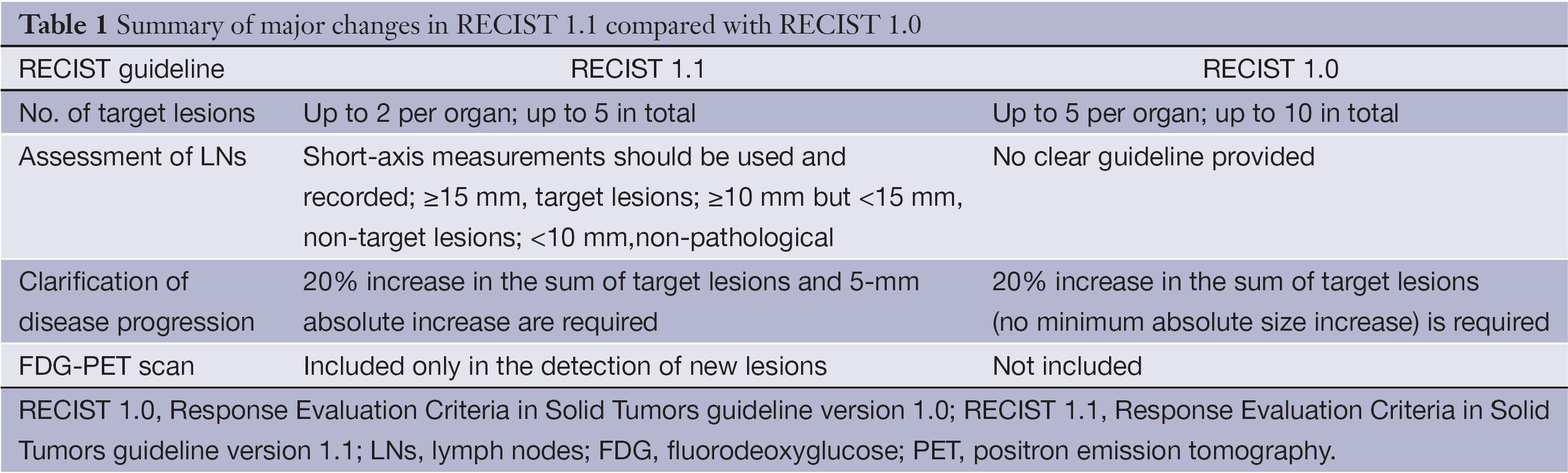
Comparison of RECIST version 1.0 and 1.1 in assessment of tumor response by computed tomography in advanced gastric cancer - Chinese Journal of Cancer Research

Table 1 from New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). | Semantic Scholar
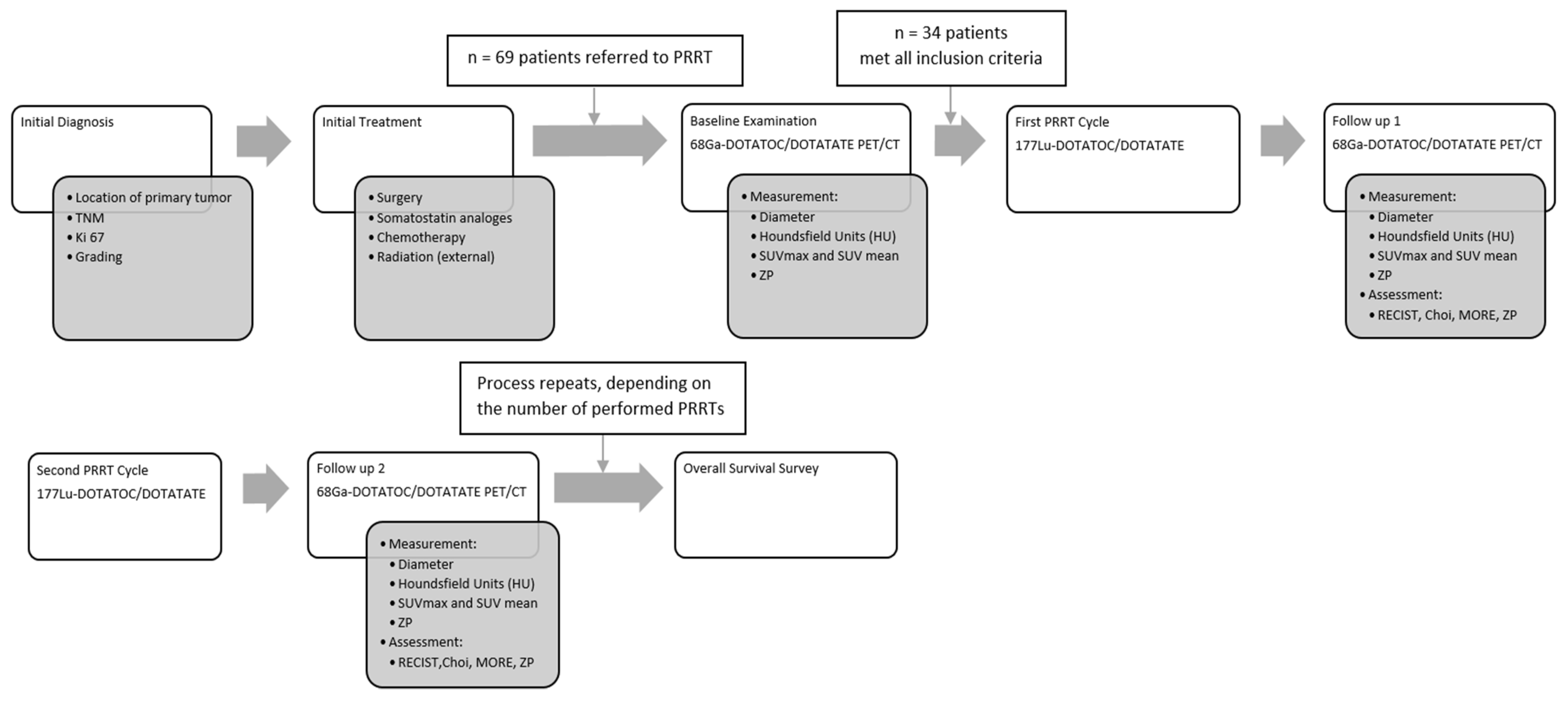
Pharmaceutics | Free Full-Text | Comparison of Choi, RECIST and Somatostatin Receptor PET/CT Based Criteria for the Evaluation of Response and Response Prediction to PRRT
Utilization of target lesion heterogeneity for treatment efficacy assessment in late stage lung cancer | PLOS ONE










:max_bytes(150000):strip_icc()/recist-5206400_final-04c81c00d8cc4583975603ca53a6ff98.jpg)